The following are the steps for submitting new application for research involving human subjects:
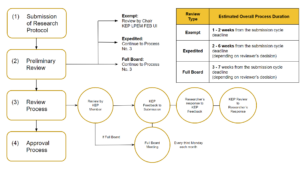
Note: This graph only illustrates the key points of KEP ethical review
1. Estimated Overall Process Duration
Referring to the Standard Operating Procedures (SOP) of KEP, this section provides an estimated duration of ethical review process from the deadline of submission cycle to the issuance of KEP decision.
The estimated duration of exempt review process is:
- ± 1 – 2 weeks from the submission cycle deadline
The estimated duration of expedited review process is:
- ± 2 weeks from the submission cycle deadline if the research is approved without modification or additional requirements
- ± 5 – 6 weeks from the submission cycle deadline if the research is approved with modification or additional requirements (with a note that investigator’s response and revision to KEP comments are considered sufficient by the reviewer)
The estimated duration of Full Board review process is:
- ± 3 weeks from the submission cycle deadline if the research is approved without modification or additional requirements or the research is rejected.
- ± 6 – 7 weeks from the submission cycle deadline if the research is approved with modification or additional requirements (with a note that investigator’s response and revision to KEP comments are considered sufficient by the reviewer)
2. Submission of Research Protocol
Application for ethical approval to KEP is eligible for all Postgraduate Students, Lecturers, and Researchers who will conduct research (whether affiliated or non-affiliated to the University of Indonesia).
The Principal Investigator or its representative is responsible for submitting the completed documents and application form along with the required attachments via email to KEP LPEM FEB UI at kep@lpem-febui.org. The following are format for the subject of email submission:
New Submission Email Subject Format
| Format:
New Application – (Name of PI) – (Name of Institution) – (Month and Year) Example: New Application – Gregory Mankiw – LPEM FEB UI – April 2021 |
Investigators can check the status of submission regularly on KEP LPEM FEB UI website: Application Status.
KEP LPEM FEB UI sets a deadline for submission every month. Every submission, whether new submission, renewal, or amendments, which are submitted until the submission deadline every month and the completion of documents will be immediately checked by the Administrator. Any submissions received after the monthly submission deadline will be proceeded to the next month. The deadline for submission is on a different date for each month because it refers to KEP Full Board meeting which will be held every third Monday of each month.
Investigators have the responsibility to check the submission deadline every month on KEP website. Researchers can access information about these important dates on the KEP website: Submission Dates and Deadlines.
KEP suggests investigators to submit documents before the submission deadline so that all corrective documents can be reviewed in the same month of submission cycle. If the information on the research protocol and documents is incomplete:
- If the investigator submits documents before the submission deadline, Administrator will ask investigator to complete the document submission and investigator has time until the submission deadline to complete the document and resubmit the completed documents to be processed in the same month of submission cycle.
- If the investigator submits documents on the submission deadline, Administrator will ask investigator to complete the document submission, but the completed and resubmitted documents will be processed in the next month of submission cycle.
3. Preliminary Review
The preliminary review consists of two stages. The first preliminary review will be conducted by Administrator to review the completion of application. If the application is incomplete, it will be returned to Principal Investigator for completion. If the application is complete, it will be forwarded to Chair of KEP. The next stage is a preliminary review by Chair to determine the type of review (exempt, expedited, Full Board) and the reviewer team. Investigators can find out the type of review for their submission on Application Status within ± 1 week from the submission cycle deadline.
4. Review Process
a. Limited Review for Certain Categories of Exemption (Exempt Review)
Exemption from review can only be granted for certain categories. The determination of the exemption status is decided by Chair and can be approved immediately if one of the certain exemption categories applies. Therefore, the research protocol with the Exempt Review category is not reviewed by KEP reviewer team and is not given feedback by KEP.
Further information regarding Limited Review for Certain Categories of Exemption (Exempt Review) can be accessed on KEP website: Exempt Categories and Expedited Review.
b. Expedited Review
Expedited review is a type of review for human subject research that involves no more than minimal risk for participating subjects.
Minimal risk means that the probability and magnitude of harm or discomfort anticipated in the research are not greater in and of themselves than those ordinarily encountered in daily life or during the performance of routine physical or psychological examinations or tests.
Further information about Expedited Review can be accessed on KEP website: Exempt Categories and Expedited Review.
c. Full Board Review
Full Board review is applied to research protocol with more than minimal risk. After review by Members, the research protocol will be brought to KEP Board at Full Board meeting. The decision to approve or reject the research protocol is determined at Full Board meeting.
5. KEP Feedback to Submission
If appropriate, KEP reviewer will give feedback to the investigator’s submission and the investigator has to respond to this feedback. Referring to the Standard Operating Procedures (SOP) of KEP, the following is an estimated time of when investigator will receive a feedback from KEP:
- Expedited: ± 2 weeks from submission cycle deadline
- Full Board: ± 3 weeks from submission cycle deadline
Investigator are given 7 working days or a maximum of 10 calendar days to write responses and revisions to KEP feedback.
6. Approval Process
If investigator’s response is not considered sufficient by KEP reviewer team, KEP review team can ask investigator to provide additional information or revision until KEP reviewer team can make a decision on the submission. Investigator will receive a notification within ± 1 week from the end of investigator’s response period if KEP reviewer team decide to ask for additional information or revision from the investigator.
If investigator’s response is considered sufficient by KEP reviewer team, then the submission can proceed to the approval process. KEP Decision Letter will be sent to the investigator within ± 1.5 weeks from the end of investigator’s response period.
KEP Decision Letter is valid for the period that is adjusted to the level of risk, but the maximum period is up to 1 year (365 days) from the issuance of the Decision Letter. Investigators also have the responsibility to remember the expiration date of KEP approval.
7. Feedback and Record-Keeping
KEP LPEM FEB UI will keep all records, including correspondence and application documents for a maximum of 5 years from the completion of human subject research.
8. Continuing Review and Renewal
KEP LPEM FEB UI will conduct a re-review of a research that has more than minimal risk of at least once every year (within 365 days from the previous approval). KEP has the authority to conduct more frequent review than that, depending on KEP’s assessment of the risk-benefit ratio of research.
Investigator have the responsibility to remember the expiration date of KEP approval. In this case, KEP will give notification if the approval is about to expire. Before the expiration of KEP approval, investigator will receive a request to complete a progress report for continuing review by KEP. Investigator have the responsibility to complete the continuing review and submit it to KEP before the approval of KEP expires. Investigator can not continue research activities if the expiration date of approval has passed and the approval for renewal has not been issued by KEP.
Further information regarding the process of renewal submission can be accessed on KEP website: Renewing Approval.


 Bahasa Indonesia
Bahasa Indonesia